Written by Jesse McLaren
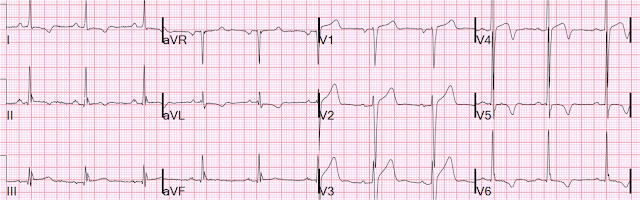
A 40 year old presented with 90 minutes of midsternal chest pressure, radiating to bilateral arms, with shortness of breath. Below is the first ECG, signed off by the over-reading cardiologist agreeing with the computer interpretation: “ST elevation, consider early repolarization, pericarditis, or injury”. What do you think?
There’s normal sinus rhythm with normal conduction, right axis and delayed R wave, and normal voltages. There’s ST elevation in V3-4 which meets STEMI criteria, which could be present in either early repolarization, pericarditis or injury. But there are also hyperacute T waves (HATW) in V4-5, which exclude early repolarization and pericarditis, leaving only LAD occlusion for this patient presenting with classic symptoms of ACS.
Here’s the PMCardio Queen of Hearts AI Model interpretation:
This is great example of how the Queen uses proportionality to identify hyperacute T waves: the T waves in V3 and V4 are almost identical, but in V3 they are proportional to its large QRS whereas in V4 they tower over its small QRS; the T wave in V5 is much smaller, but relative to its QRS it is large and bulky.
So using the OMI paradigm and its AI, this patient would have had immediate cath lab activation before the first troponin result. Let’s see what happens in the current STEMI paradigm.
Emergency physician: ‘STEMI neg’ but with elevated troponin = Non-STEMI
The first ECG was signed off. After only 90 minutes of chest pain, the first troponin was unsurprisingly in the normal range at 11ng/L (normal <26 in males and <16 in females), so the emergency physician waited for repeat troponin. After it rose to 150ng/L two hours later a repeat ECG was done:
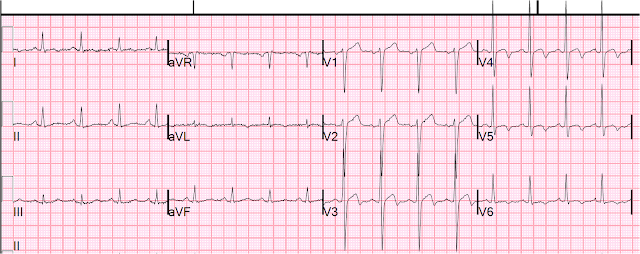
There’s now a Q wave in V3 and a smaller T wave in V3-V4, proving this is LAD occlusion. Hyperacute T-waves remain in V3 and V4. There’s the same degree of ST elevation, but this time the computer calls it STEMI. But it was interpreted as no acute ischemia and the patient was referred to cardiology as Non-STEMI.
Nurse notes: the silent scream of the heart
The emergency nursing notes document the patient complaining of chest pain refractory to nitro, with a rising trop:
2200: ECG shown to ED MD
0020: repeat ECG shown to ED MD, patient complain of midsternal chest pain
0520: nitro x 3. Chest pain still persists. Cardiology aware. Repeat troponin ordered
0630: lab called for high troponin 3900. Paged cardiology
0800: patient complains of chest pain. Repeat blood work and ECG
0845: repeat trop over 7000. Cardio aware
1030: repeat trop sent, no change in chest pain
1100: heparin drip started
1130: transfer for cath, still complains of chest pain
Here’s the ECG repeated at 0800
There’s now Q waves V3-4. T waves have deflated and inverted in V3-4 suggesting some degree of reperfusion, but the patient still complained of ongoing chest pain. Troponin rose to 12,000 before cath.
Cardiology: delayed cath = Non-STEMI
Here’s the interventional cardiology note describing the ECGs, management and outcome:
“He has had transitory peak T-waves, ST-segment elevation, and biphasic T-waves during recurrences of pain. This morning, he was also found to have a rising troponin. He was thus referred for emergent invasive assessment. Code STEMI was called…Mid LAD had serial 70 and 60 percent lesions and was occluded in the distal segment…An excellent result was achieved. The total occlusion was recanalized and stented from 100 to 0%.”
Here's the discharge ECG, with ongoing Q waves and reperfusion T wave inversion:
Peak troponin was 47,000 ng/L and echo showed an akinetic apex with EF 45%.
What should the discharge diagnosis be, and why does this matter?
The patient had an ECG with ST elevation (and hyperacute T waves), activation of code STEMI, a 100% LAD occlusion on angiogram, a massive peak troponin, and an akinetic wall. The discharge diagnosis should reflect the underlying pathology of Occlusion MI, regardless of whether the ECG was interpreted to show STEMI criteria, and regardless of time to reperfusion. But because of the delayed reperfusion, the discharge was Non-STEMI.
Thus, a case with more than 12 hours of delay for reperfusion will not be flagged for review.
_______
Smith: we have an article under review that shows that the variable most closely associated with the final diagnosis of "STEMI" vs. "Non-STEMI" was a door to balloon time less than, vs. greater than, 90 minutes. Not whether there was or was not Acute Coronary Occlusion. Not whether the ST segments met STEMI millimeter criteria.
_______
The problem is not just for this patient, and simply changing the discharge diagnosis in this case from "Non-STEMI" to "STEMI" is not the solution. This is just an example of the broader problems of the STEMI paradigm for research and quality improvement.
For research, 12 hours to PCI is deemed ‘early intervention’ for Non-STEMI. If two of the same patients were part of Non-STEMI trials like TIMACS, and randomized into ‘early’ (16 hours) vs ‘delayed’ intervention (52 hours), there would appear to be no benefit to the ‘early’ intervention, because the damage was already done. But clearly this 'Non-STEMI' patient with OMI would have benefited from immediate cath lab activation on arrival, when their first troponin was 11ng/L, rather than after after it rose to 12,000ng/L after 12 hours of refractory ischemia.
For quality improvement, the discharge diagnosis also matters. Classifying as STEMI vs Non-STEMI can be more reflective of reperfusion time than ECG findings or patient outcomes, which allows cases like these to be normalized. Instead, if patients are classified by the actual outcome of OMI vs NOMI, then this patient clearly had a missed OMI. This is not to assign blame but to identify multiple opportunities for improvement:
1. ECG: using OMI signs and AI, to activate the cath lab on arrival, before waiting for the troponin
2. POCUS for complementary regional wall motion abnormalities for subtle OMI
3. Clinical: patient alerts for refractory ischemia (refractory chest pain), and empowering nurses to advocate for patients
4. Troponin: troponin is a rear-view mirror that shows damage that has already happened, so the first troponin is unreliable with acute symptoms and serial troponin will lag behind the damage of ongoing occlusion. But refractory ischemia with rising troponin is an indication for cath lab activation regardless of the ECG.- i) The errors of omission and commision in today's case are multiple and preventable.
- ii) Since it is now 7 years since initial publication of the OMI Manifesto (See the April 18, 2018 post in Dr. Smith's ECG Blog) — We would have expected that emergency providers (especially cardiologists!) would by now accept the expanding body of literature demonstrating that at least 1/3 of all acute coronary occlusions are missed by clinicians "stuck" on the outdated millimeter-based STEMI protocol (our every expanding OMI Literature Timeline — being readily accessible in the upper Menu Bar Tab at the top of each page in this ECG Blog).
- iii) Cardiologists are all-too-often rewarded for misclassifying all-too-many MIs as a "NSTEMI" — often in cases in which there is foolproof evidence of acute coronary occlusion that should have been recognized many hours earlier. Take today's case, in which Dr. McLaren highlights how despite more than 12 hours of delay for reperfusion — the interventionist note indicates "an excellent result was achieved". Especially concerning is that for data-keeping (research) purposes — this 12+ hour delay until PCI was finally achieved, will end up being viewed not as an unfortunate delay — but as a beneficial "early" intervention by the irony that this event was misclassified as a "NSTEMI".
- MY Thoughts: While fully acknowledging that I viewed today's initial ECG in the comfort of my home office in front of my large screen computer — there are findings on this initial ECG, that in a patient who presents with new-onset severe CP have to be recognized!
-USE.png) |
| Figure-1: Today's initial ECG. |
- There is no need to wait for Troponin results — and not even any need to repeat the ECG prior to activating the cath lab.
- And, as soon as the decision is made to activate the cath lab — IV morphine could have been given to relieve this patient's chest pain (that the patient had to experience for more than 12 hours).
- After identifying that the initial rhythm in Figure-1 is sinus — my "eye" was immediately drawn to the hyperacute T wave in lead V4 (within the RED rectangle). Although true that some repolarization variants may be marked by tall, peaked T waves — the disproportionate enlargement of this T wave in lead V4 (that is no less than 13 mm tall!) in this patient with severe new-onset CP clearly exceeds the dimensions reasonably expected for a repolarization variant.
- In the context of this hyperacute T wave in lead V4 — the T wave in neighboring lead V5 has to be also interpreted as hyperacute (clearly "bulkier"-than-it-should-be given modest size of the R wave in this lead).
- Any doubt that may have existed about the acuity of these lead V4,V5 findings — should be alleviated on seeing the ST-T wave in lead aVL (While the T wave inversion in aVL may sometimes be a normal finding when the QRS is predominantly negative — there should not be ST segment coving with slight elevation in this lead as we see in ECG #1).
- As per Dr. McLaren, although the T wave in lead V3 is equally tall as the T wave in lead V4 — it is associated with a much deeper S wave. In the context of ST-T wave appearance in the other 5 chest leads — I still interpreted this T wave in lead V3 as hyperacute, but I would have been less certain of that interpretation if other chest leads were normal.
- In the context of ST-T waves in leads V3,V4,V5 — I interpreted the coved and elevated ST segment in lead V2 as an acute change (I thought the amount of J-point elevation in lead V2 to be more than is normally seen in this lead).
- Similarly — I interpreted the ST segment coving and slight elevation in neighboring lead V1 as abnormal (while fully acknowledging that as an isolated finding — I would not interpret the ST-T wave appearance in lead V1 as abnormal).
- Finally, in the context of the other 5 chest leads — I thought the T wave in lead V6 to be "fatter"-at-its-peak and wider-at-its-base than I would normally expect given the modest R wave amplitude in this lead.
- QRS morphology in lead I is that of an rS complex.
- QRS morphology in leads II,III,aVF is that of a qR complex.
- This QRS morphology in these 4 limb leads is completely characteristic of LPHB (Left Posterior HemiBlock). Although it is rare to see LPHB as an isolated conduction defect (LPHB almost always occurs as a bifascicular block, in association with RBBB) — this patient's ECG is otherwise diagnostic of acute LAD OMI, so this could be a new conduction defect occurring in association with acute LAD OMI (ie, There is no baseline ECG to compare this limb lead morphology to — so we cannot tell if this is or is not a new conduction defect).
- Because the left posterior hemidivision is typically a thick, diffuse fascicular bundle (as opposed to the much thinner, and much more easily injured left anterior hemifascicle) — acute LPHB is a potentially serious conduction disorder (that potentially might need pacing if this LAD OMI is not treated in a timely manner).
- Failure to recognize potentially new LPHB is yet one more oversight in today's case.



















-USE.png)